This site uses cookies for analytics and to improve your experience. By clicking Accept, you consent to our use of cookies. Learn more in our privacy policy.
Open-heart surgery is a high-precision procedure, yet the immediate postoperative period remains a critical window. Recovery depends on the ability of chest tubes to stay clear and drain residual blood effectively. Recent clinical guidelines from EACTS emphasize that effective chest tube drainage is essential for patient recovery1. When these tubes obstruct—which occurs in as much as 36% of patients2—the consequences are severe: excessive bleeding, retained blood syndrome, and the high-risk necessity of surgical re-exploration3, 4.
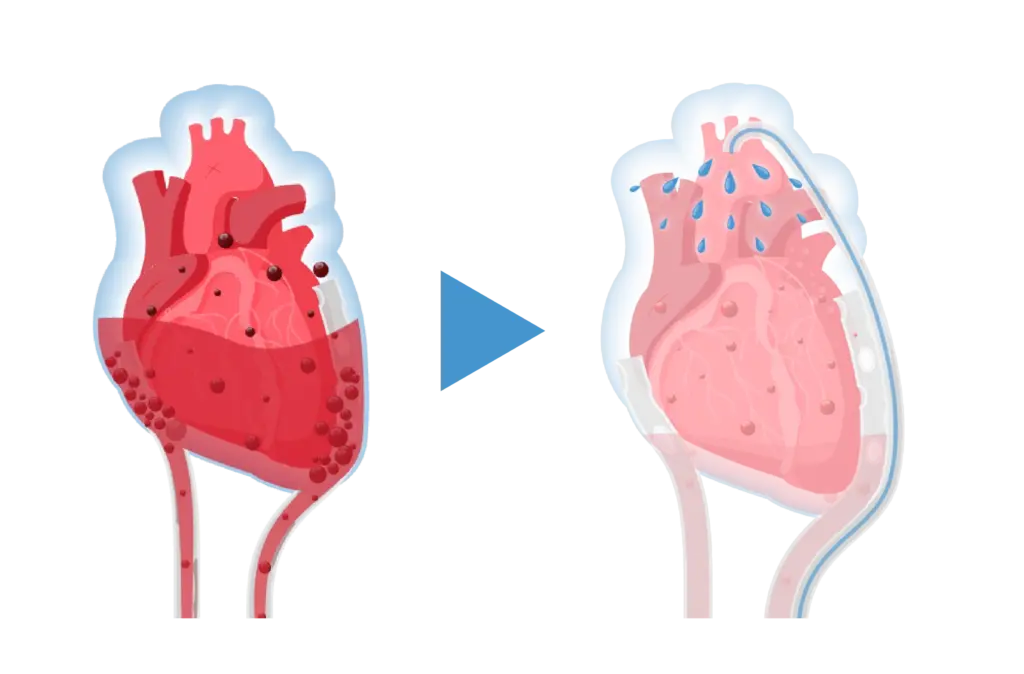
Traditionally, a reoperation is the only way to clear these clots—a reactive process of reopening and rinsing the chest. However, evidence highlights that surgical re-exploration for bleeding is a major independent predictor of adverse outcomes, significantly increasing 30-day mortality4. The Haermonics Pure System addresses this challenge by targeting the problem at its source.
David R. Koolbergen, M.D. Ph.D.
Inventor of the therapy
Congenital Cardiothoracic Surgeon at Amsterdam UMC / Leiden UMC

2013
1 pilot trial6
2 RCTs7-8
References
[1] Casselman, F. P. A., et al. 2024 EACTS/EACTAIC Guidelines on patient blood management. European Journal of Cardio-Thoracic Surgery. 2024. 67(5).
[2] Karimov JH, et al. Incidence of chest tube clogging after cardiac surgery: a single-center prospective observational study. Eur J Cardiothorac Surg. 2013.
[3] Niemann B, Grieshaber P. Retained blood syndrome after cardiac surgery. Eur J Cardiothorac Surg. 2025.
[4] Čanádyová J, et al. Re-exploration for bleeding or tamponade after cardiac operation. Interact Cardiovasc Thorac Surg. 2012.
[5] Klautz, M. J. (2024, October). Clinical impact of continuous postoperative pericardial flushing on bleeding-related complications after cardiac surgery: A pooled analysis. [Oral Presentation]. 38th EACTS Annual Meeting, Lisbon, Portugal.
[6] Manshanden JS, et al. Continuous postoperative pericardial flushing: a pilot study on safety, feasibility, and effect on blood loss. EBioMedicine. 2015.
[7] Diephuis E, et al. Continuous postoperative pericardial flushing method versus standard care for wound drainage after adult cardiac surgery: a randomized controlled trial. EBioMedicine. 2020.
[8] Diephuis E, et al. Continuous postoperative pericardial flushing reduces postoperative bleeding after coronary artery bypass grafting: a randomized trial. EClinicalMedicine. 2020.
[9] Molenaar MA, et al. Effects of Continuous Postoperative Pericardial FLUshing with Investigational Device on Postoperative Re-Explorations for Bleeding (FLUID)—Randomized Clinical Trial. J Clin Med. 2026; 15(6): 2151.
[10] Dyke C, et al. Universal definition of perioperative bleeding in adult cardiac surgery. The Journal of Thoracic and Cardiovascular Surgery. 2014;147(5):1458-1463.
[11] Batchelor TJP, et al. Guidelines for Perioperative Care in Cardiac Surgery: Enhanced Recovery After Surgery (ERAS) Cardiac Society Recommendations. JAMA Surgery. 2026.

Share your email address with us and we’ll share important milestones and developments with you.
Since 1875, cardiac drainage methods have remained largely unchanged despite the persistent risk of post-operative complications. At Haermonics, we didn’t just think we could improve this standard—we proved it.
As a clinical-stage medtech company, we developed the Haermonics Pure System, an innovative flush therapy designed to proactively prevent post-operative cardiac surgery complications. By maintaining clear drainage, we eliminate the guesswork and risks associated with traditional methods.
Since 1875, cardiac drainage methods have remained largely unchanged despite the persistent risk of post-operative complications. At Haermonics, we didn’t just think we could improve this standard—we proved it.
As a clinical-stage medtech company, we developed the Haermonics Pure System, an innovative flush therapy designed to proactively prevent post-operative cardiac surgery complications. By maintaining clear drainage, we eliminate the guesswork and risks associated with traditional methods.
Our team integrates a wealth of experience across the entire medical device lifecycle, bringing together specialized expertise in several domains. By combining these diverse roles, we ensure that our innovation is not only technically superior but also rigorously validated and prepared for global clinical adoption.
Our strategic direction is further refined by a diverse Advisory Panel that provides a 360-degree view of the cardiac surgery ecosystem. Their combined wisdom ensures the Haermonics Pure System addresses the specific clinical and economic needs of surgeons, intensive care units, hospital administrations, and payers alike.
















Haermonics Head office
Microlab
Kastanjelaan 400
5616 LZ Eindhoven
The Netherlands
Would you have 1 minute (literally) to answer 3 reseach queries we have?